If crystals of a relatively insoluble substance are placed in contact with a saturated solution of the substance, an exchange of ions between the solution and surface will take place. If the ions in solution are labeled with a radioactive isotope, this exchange, which depends upon the surface area of the crystals, can be determined, since at equilibrium:
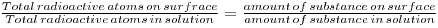
If the molecules are assumed to be cubic:
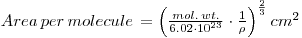
where ρ is the denisity (6.2 g/cm3 PbSO4).
Since there will be 1 radioactive atom per molecule, the specific surface area (i.e. the surface area per gram of sample) can readily be calculated.